No unexpected COVID-19 vaccine safety issues have been identified in Canada to date according to Health Canada. That comes in light of Wednesday’s updated product label for the AstraZeneca and COVISHIELD vaccines, reflecting rare reports of blood clots and low platelet counts.
In a press conference on Thursday, Dr. Howard Njoo, Canada’s Deputy Chief Public Health Officer, said while there have been some reports of blood clots in the European Union, so far, the serious events are very rare.
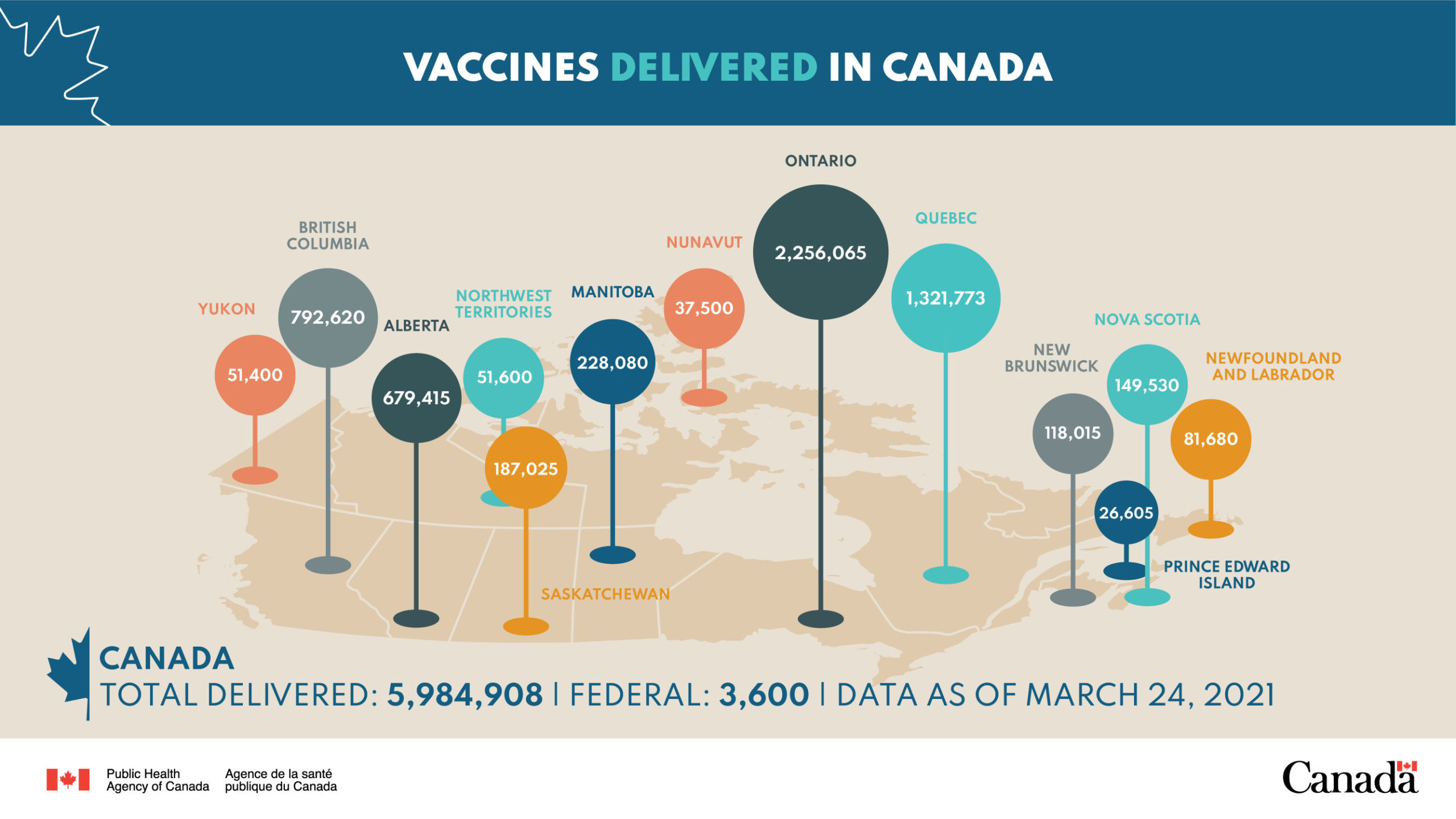
Dr. Njoo said that to date, provinces have administered over 300,000 doses of the COVISHIELD-AstraZeneca vaccine with no reports of blood clots in the country.
Over 4.3-million doses of COVID-19 vaccines have been administered nation-wide as of Thursday. Dr. Njoo said more than 11% of eligible Canadians over the age of 18 have received at least one dose- including 60% of people over 80 years old, 19% of those aged 70 to 79 years and more than 60% of adults in all three territories.
Canada is also set to receive over 16-million vaccines between April and June.
Major General Dani Fortin, the head of Vaccine Logistics, confirmed over 12-million vaccines will be arriving from Pfizer-BioNTech alone. Fortin said this week’s deliveries of 846,000 Moderna doses were divided into two shipments. One is currently being delivered to provinces and territories and the other is expected to arrive Saturday for distribution next week.
According to Fortin, Moderna is set to deliver 855,000 doses the week of April 5th and an estimated 1.2-million the week of April 19th. He also confirmed the 1.5-million AstraZeneca doses from the U.S. are expected to arrive next week.
In addition, the Serum Institute of India is set to deliver over 1-million AstraZeneca doses in April and 500,000 doses in May.